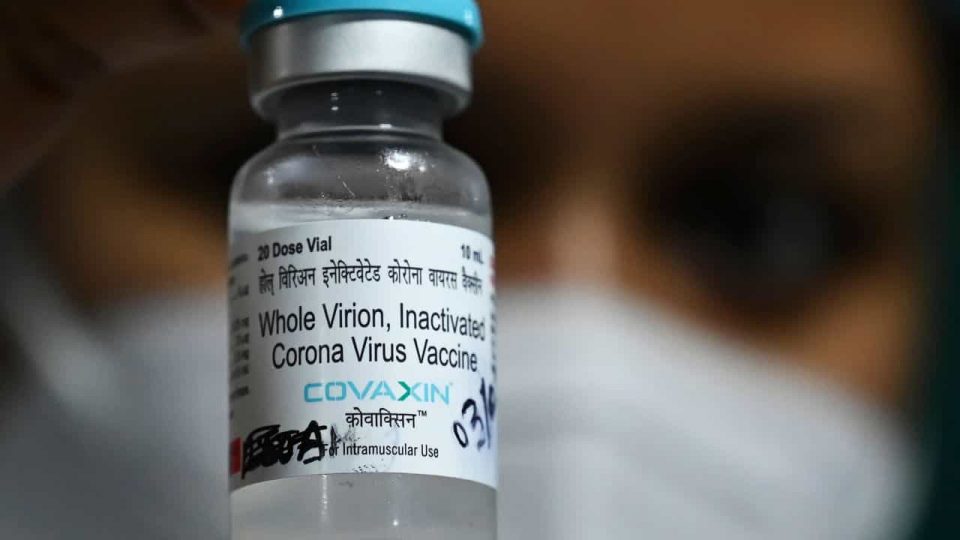
As the threat of the third wave of Covid-19 appears large over India, the AIIMS Chief expects approval of the Covaxin coronavirus vaccine for children by October. AIIMS Director Dr Randeep Guleria said that Bharat Biotech is expected to submit clinical trials on children by September-October. The pandemic can be controlled only if everyone is vaccinated, he said.
“Bharat Biotech and other companies are doing trials at a breakneck speed as parents have come forward with their children for the trials,” he said. “One is hopeful that the trial will be completed early and possibly with follow up of about two-three months, we shall have data by September.
- Godrej Properties Shares Zoomed 9.5% on Robust Q4FY24 Business and Financial Performance
- Stocks in Focus: Titan, Aurobindo Pharma, Britannia Industries, and Others
- Stocks Under F&O Ban: Balrampur Chini, Aditya Birla Fashion, Biocon, and Others
- How to choose the Best Stocks for Intraday F&O trading in India?
- Vedanta Outlook for the Week (May 06, 2024 – May 10, 2024)
Hopefully, by that time, vaccines will be approved so that by September-October, we can give to children,” he added.
He further said that the Covaxin “covers a wide spectrum” as Bharat Biotech conducts trials between the age group of 2 to 18 years. Covaxin, developed by Bharat Biotech in collaboration with the Indian Council of Medical Research (ICMR), is being used on adults in India’s current vaccination drive.


 Signals, Powered By EquityPandit
Signals, Powered By EquityPandit