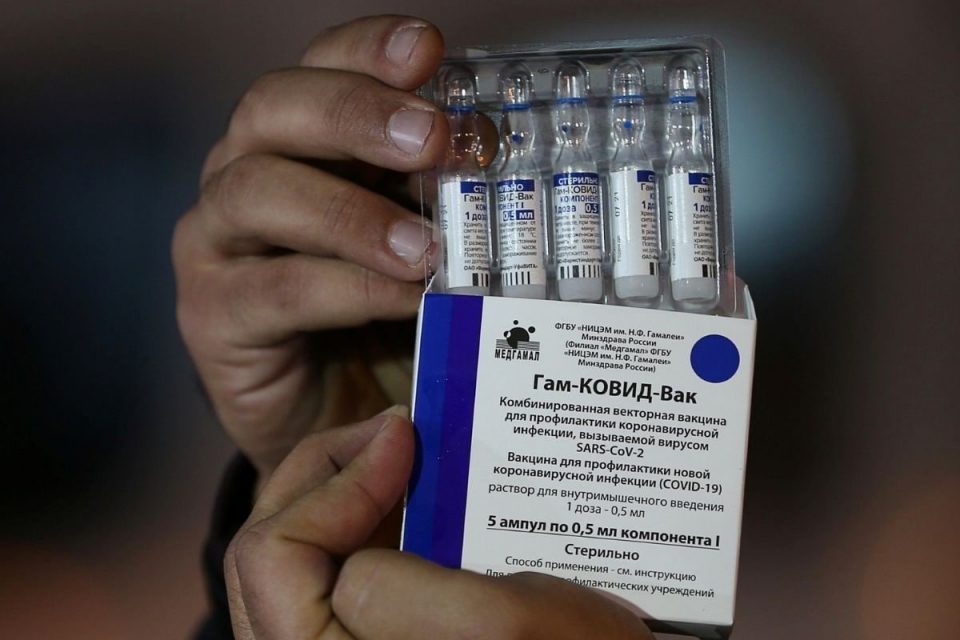
The Drug Controller General of India (DCGI) has approved the restricted emergency use of the Sputnik V vaccine late Monday night. With this, India now has three vaccines against COVID-19, including Covishield the Oxford-AstraZeneca vaccine manufactured by the Serum Institute of India, and Bharat Biotech’s Covaxin According to a release issued by the Russian Direct Investment Fund (RDIF), India is the most populated country to register the Russian vaccine. “Total population of 60 countries where Sputnik-V is approved for use is 3 billion people or about 40% of the global population,” it added.
- Infosys Announced Business and Financial Updates of Q4FY24, Shares Traded Flat
- Rivos Startup Raised USD 250 Million To Build Silicon Chip for AI Models, Powering Data Analytics Applications
- Crude Oil Prices Jumped 3% Following the Recent Israel Attacks on Iran
- ITC Stocks Gained 1.4% on Arm to Acquire 100% Stake in Blazeclan Technologies
- Indus Towers Shares Hit 52-Week High on Signing MoU for Renewable Energy Projects
The vaccine has been registered in India under the emergency use authorization procedure based on results of clinical trials in Russia as well as positive data of additional Phase III local clinical trials in India conducted in partnership with Dr. Reddy’s Laboratories, it added. “India is the leading production hub for Sputnik-V. RDIF has reached agreements with the leading pharmaceutical companies in the country (Gland Pharma, HeteroBiopharma, Panacea Biotec, Stelis Biopharma, Virchow Biotech) aimed at the production of more than 850 million doses per year,” noted the release.


 Signals, Powered By EquityPandit
Signals, Powered By EquityPandit