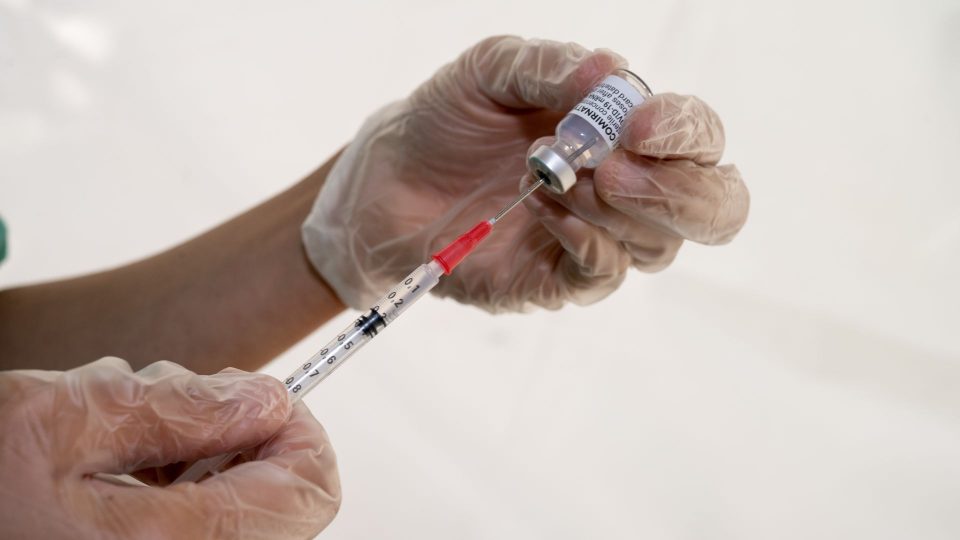
The Food and Drug Administration (FDA) approved the Covid-19 vaccine booster for people with weak immune systems. “Today’s action will allow doctors to boost immunity in weak immune systems who need extra security from coronavirus,” FDA Commissioner Dr Janet Woodcock said in a statement.
The agency said people who are similarly immunocompromised could take a third dose of the covid vaccines. They also highlighted that fully vaccinated people are “sufficiently protected” and do not need an extra amount. The Centers for Disease Control and Prevention’s vaccine advisory committee has scheduled a meeting to study vaccine boosters for immunocompromised peoples. If it is approved, the third shot can begin immediately.
- Kotak Mahindra Bank Shares Crash 13% Post RBI Action
- Nestle India Joins Hands With Dr Reddy for Nutritional Health Solutions
- Inox Wind Shares Traded 9% Higher On Board Proposal For Bonus Shares Issuance
- Nestle India Stocks Gained 3% on Q4FY24 Business and Financial Updates
- Rail Vikas Nigam Shares Gain 3% on Securing Project Worth Rs 239 Crore
White House Chief Medical Advisor Dr Anthony Fauci said that federal health officials are advancing regulatory efforts to clear 3rd doses for people who have cancer and HIV or need organ transplants. He told new data advised they don’t generate sufficient immune even after taking two doses of Covid-19 vaccine. “Immunocompromised peoples are unsafe. Hence, we need to get those people vaccine boosters, and we are working on that, and it will be implemented as quickly as possible.” Fauci said.


 Signals, Powered By EquityPandit
Signals, Powered By EquityPandit